Huntington’s Disease; Gene Silencing could turn off Harmful Gene Mutation
Posted by Phil Heler, MD on May 3, 2019Huntington's Disease is found in 1 in 10,000 people in Western Europe and it is caused by a gene mutation
Huntington’s Disease is an important neurological condition. Why write about is now? As Easter comes and goes there are other noteworthy landmark anniversaries worth celebrating this month. April 25th is the anniversary of when Watson and Crick discovered the double helix structure that comprises our DNA in 1953. Also, by coincidence in the same month, on April 14th (2003) the Human Genome Project was also completed. Therefore, it feels entirely appropriate to discuss the legacy of these pioneering scientific events.
What can DNA Tell us?
Clearly things have moved on rapidly since these defining moments. DNA tests now have all sorts of applications from establishing the perpetrator of a criminal act, the gender of a baby, paternity tests (watch out Justin Beiber!) or to establish our ancestral origins or our potential for ill health.
It has also been the topic of popular culture; the author Michael Crichton wrote a cautionary tale about genetic engineering that so captured our imaginations in the movie ‘Jurassic Park’. It is now possible for any of us to order an online direct to consumer genetic testing kit. From this we can trace our origins and determine if we have harmful variants of any given gene. Indeed, our origins are something that we are all fundamentally fascinated with. This has been reflected by popular TV shows such as ‘Who Do You Think You Are?’.
This fascination that we have with our genealogies has also provided business opportunities for rapidly expanding companies such as Ancestry.com. Our genes of course do also offer useful information about our potential for ill health. Your very own version of any given gene will confer an increased or lowered predisposition for certain outcomes. Obesity is an interesting case as half the population in the UK carry a variant of the FTO gene which will make them 1.6 kg heavier than the rest of the population. 16% of the population carry two copies of the FTO gene which will make them on average 3 kg heavier.
However, it is worth pointing out, this is also influenced by your environment, how much you eat and exercise. On a more detailed basis we now know that at least four genes work together to determine eye colour, and this can be predicted with 97% certainty. Even our internal body clock can be predicted. If we have a Per2 gene, we are likely to go to bed early and get up early and if we have the Per3 gene we are more likely to be a night owl. Versions of the Klotho gene meanwhile have been associated with how long we live!
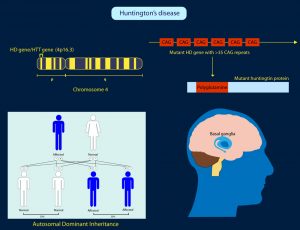
A host of genetic tests can be performed for rare disorders. These can determine our predisposition for diseases such as diabetes, cancer, Alzheimer’s or cystic fibrosis. Huntington’s disease for instance is found in 1 in 10,000 people in Western Europe and it is caused by a mutation in the Huntington gene. We each carry two copies of this gene and only one needs to be defective. These predictive tests essentially enable the identification of a genetic mutation before a disease process occurs. We now know for instance that up to 80% of women with the BRCA1 or BRCA2 genes will develop breast cancer. It is even possible to work out if your children are likely to inherit a harmful variant of a gene
What is DNA?
It was back in 1953 that James Watson and Francis Crick described the double helix structure of deoxyribonucleic acid (DNA), the chemical compound that contains the genetic instructions for building, running and maintaining living organisms. DNA in each of us hosts about 21,500 genes (collectively known as a genome) that are spread into the ladder-like structure of a DNA molecule. To give you an idea of the shear complexity of all this, a DNA structure has roughly 3 billion rungs in its ladder where the genes are coded. DNA is simply comprised of four chemical bases called adenine, thymine, cytosine and guanine (or A, T, C and G). Pairs of these (called nucleotides) form the “rungs” of our twisted, ladder-shaped DNA molecules.
All genes are made up of stretches of these four bases, arranged in different ways and in different lengths. The Human Genome Project, which took 12 years, finally finished decoding all these genes in 2003 and gave detailed information about the structure, organisation and potential function of our entire genome. We now have a complete map to what can be considered as the basic set of inheritable instructions for our development and function. Francis Collins the director of the National Centre for the Human Genome Project (NCHGP) back in 2003 reflected on the huge achievement by stating, “It’s a history book – a narrative of the journey of our species through time. It’s a shop manual, with an incredibly detailed blueprint for building every human cell. And it’s a transformative textbook of medicine, with insights that will give health care providers immense new powers to treat, prevent and cure disease.”
The Human Genome Project’s key goal was to provide researchers with powerful tools to understand the genetic factors in human disease, paving the way for new strategies for diagnosis, treatment and prevention. Of course, the key to all this is learning what each specific gene does. Having the complete sequence of the human genome is a bit like having all the pages of a user manual to make a human body. Our challenge now is to determine how to read the contents of these pages. We then have to work out how all these many complex parts combine together to dictate human health and disease. Overall the Human Genome Project has already fuelled the discovery of more than 1,800 disease genes. As this technology moves forward one of the exciting and interesting areas of investigation is what is known as ‘gene silencing’.
Gene Silencing and Huntington’s Disease
Craig Mello and Andrew Fire, now at Stanford University School of Medicine in California, shared a 2006 Nobel Prize for discovering the process of silencing genes. Cells do naturally turn their genes on and off as a natural process. Cells undertake this process through what is called RNA interference (or RNAi) and scientists have evolved methods of tricking a cell into shutting down a specific gene. Like DNA, RNA is a molecule found in all cells. One of the jobs of RNA is to read the instructions provided by DNA and essentially act as a photocopier. So, for example, when a cell needs to make a kind of protein, it activates a particular gene in its DNA.
It then produces multiple copies of that piece of DNA in the form of messenger RNA (or mRNA). All these copies of mRNA are then used to translate the genetic code into protein through the cell’s manufacturing machinery (or ribosomes). Clearly cells in the body each have different tasks to perform, so a cell will only use certain genes at any given time depending on what it needs to do. So, a cell in your skin will require access to very different genes than, for example, a neuron in your brain. RNA molecules help this process of selecting which genes to use by using proteins called Argonauts (yep…as in Greek mythology!). These bind to the RNA and help coordinate the process by turning unwanted genes off. Clever stuff!
Latest Research Offers Hope for Huntington’s Disease Sufferers
A recent phase 1 trial, using gene silencing, on Huntington’s disease was completed in 2017. The results of this trial were hailed as enormously significant because this was the first time any drug demonstrated an ability to suppress the effects of the Huntington’s mutation gene variant that causes irreversible damage to the brain. The mutant Huntington’s gene contains instructions for cells to make a toxic protein, called huntingtin.
This code is copied by an RNA messenger molecule and dispatched to the cell’s protein-making machinery. The drug, Ionis-HTTRx, works by intercepting the RNA messenger molecule and destroying it before the harmful protein can be made, effectively silencing the effects of the mutant gene. This caused ripples of excitement across the scientific world because the drug, which was a synthetic strand of DNA, could potentially be adapted to target other incurable brain disorders such as Alzheimer’s and Parkinson’s. The Swiss pharmaceutical giant Roche has paid a $45 million licence fee to take the drug forward to clinical use.
The trial involved 46 men and women with early stage Huntington’s disease in the UK, Germany and Canada. The patients were given four spinal injections one month apart and the drug dose was increased at each session; roughly a quarter of participants had a placebo injection. After being given the drug, the concentration of harmful protein in the spinal cord fluid dropped significantly and in proportion with the strength of the dose.
This kind of closely matched relationship normally indicates a drug is having a powerful effect. Prof Sarah Tabrizi, director of University College London’s Huntington’s Disease Centre who led the phase 1 trial said, “For the first time a drug has lowered the level of the toxic disease-causing protein in the nervous system, and the drug was safe and well-tolerated. This is probably the most significant moment in the history of Huntington’s since the gene [was isolated].”
The exciting thing about this trial was that in principle any protein could be targeted. For instance, a similar synthetic strand of DNA could be made to target the messenger that produces misshapen amyloid or tau proteins in Alzheimer’s. However, for the moment this breakthrough could also be very exciting news for people who suffer from Huntington’s disease. About 10,000 people in the UK have the condition and about 25,000 are at risk. Most people with Huntington’s inherited the gene from a parent, but about one in five patients have no known family history of the disease.
The Human Genome Project has also enabled other ambitious new initiatives such as the Cancer Genome Atlas which aims to identify all the genetic abnormalities seen in 50 major types of cancer. As the example on Huntington’s demonstrates, we are beginning to have a deeper understanding of disease at the genomic level. There also is a possibility in the future for an individualised analysis, based on each person’s genome, that could help lead to a powerful form of preventive, personalised and pre-emptive medicine. There are of course some ethical dilemmas as well that go hand in hand with information.
As a society, we also will be confronted with the ethical issues that often occur when diagnostic tools are turned into discriminatory instruments for screening people or when therapeutic techniques are applied to the enhancement of individual capabilities or traits.